Trends in Oncology
How to Use PDX Models to Advance Folate Receptor-Alpha ADC Development in Ovarian Cancer
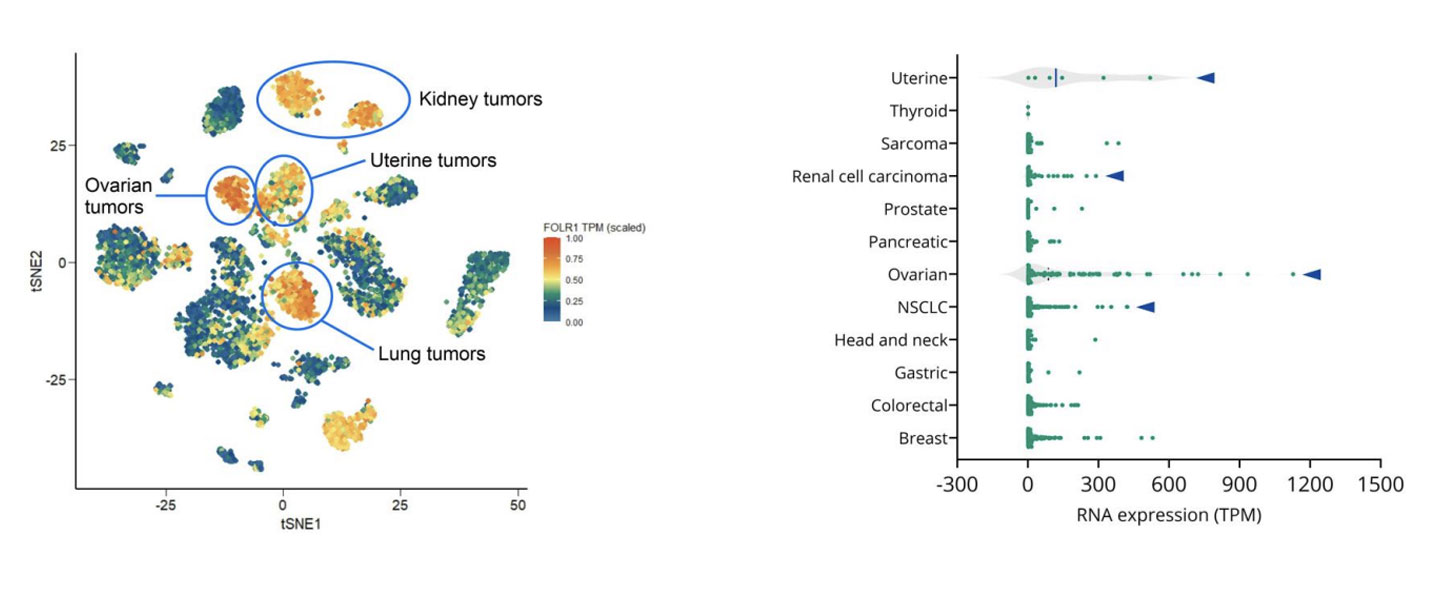
Epithelial ovarian cancer remains one of the deadliest gynecologic malignancies. Although many patients initially respond well to platinum-based chemotherapy, relapse is common, and outcomes for platinum-resistant disease have seen little improvement over the past decade. With limited treatment options and a shortage of robust predictive biomarkers, there is a clear need for more precise therapeutic strategies that can better match patients to targeted therapies. One promising area of progress is antibody-drug conjugates, or ADCs. These therapies are designed to deliver potent cytotoxic agents directly to tumor cells by targeting tumorassociated antigens. In ovarian cancer, folate receptoralpha, or FRα, has emerged as an especially compelling target due to its high expression in ovarian tumors and comparatively limited expression in normal tissues. At AACR 2026, we shared new data showing how patient-derived xenograft, or PDX, models can be used to evaluate FRα-targeted ADCs, explore variability in response, and begin to uncover potential mechanisms of resistance. This work highlights how clinically relevant PDX models, combined with molecular profiling, can support smarter ADC development decisions earlier in the pipeline. Why FRα is an attractive ADC target in ovarian cancer FRα is a transmembrane glycoprotein involved in folate transport and cellular metabolism. While healthy tissues show limited FRα expression, ovarian tumors, particularly high-grade serous carcinomas, often express it at high levels. This contrast makes FRα well suited for ADC approaches that aim to maximize tumor-specific activity while minimizing offtarget toxicity. Mirvetuximab soravtansine-gynx, marketed as Elahere®, is an FRα-targeted ADC recently approved for patients with FRα-positive, platinum-resistant ovarian cancer. However, clinical experience has shown that FRα expression alone does not reliably predict which patients will respond to treatment. Understanding this response heterogeneity is one of the key challenges in optimizing ADC development. How we select the right PDX models to reflect patient biology To address this challenge, we leveraged our gynecologic cancer PDX collection, which includes models derived from both treatment-naïve and heavily pretreated ovarian cancer patients. Using integrated RNA sequencing and immunohistochemistry, we assessed FRα expression across ovarian PDXs and compared these patterns with patient data. Consistent with clinical observations, ovarian cancer PDXs showed some of the highest FOLR1 expression levels across tumor types. Importantly, RNAbased expression data strongly correlated with membrane-specific FRα protein expression. This confirms that multiple complementary methods can be used to select biologically appropriate models for FRαtargeted ADC screening. Rather than relying on a single cutoff, this approach allows us to build PDX cohorts that better reflect the biological diversity seen in the clinic. What PDX efficacy studies reveal about ADC response Selected ovarian PDX models were treated with mirvetuximab soravtansine using a clinically relevant dosing schedule. As expected, models lacking FRα expression did not respond to treatment, reinforcing the importance of FRα as a prerequisite for activity. Among FRα-positive models, however, responses varied widely. Some tumors showed meaningful growth inhibition, while others demonstrated limited or no response despite high FRα expression. This mirrors what is seen in the clinic, where only a subset of FRαpositive patients achieves durable benefit from treatment. These results highlight a critical lesson for ADC development. Target expression is necessary, but it is rarely sufficient on its own. Using molecular analysis to explore resistance mechanisms To begin understanding why FRα-positive tumors respond differently, we compared responding and non-responding PDX models using transcriptomic and pathway-level analyses. Rather than focusing on single genes, we looked at broader biological processes that may influence sensitivity or resistance to ADC therapy. Clear differences emerged between sensitive and resistant tumors. In particular, pathways related to intracellular transport and drug efflux were enriched in non-responding models. Several of these pathways involve ATP-binding cassette transporters, which are well known for their role in multidrug resistance. While exploratory, these findings suggest potential mechanisms by which tumor cells may evade ADC-delivered payloads, even when the target antigen is present. This type of insight can help guide biomarker development and inform rational combination strategies. How we model acquired resistance using PDXs Primary resistance is only part of the challenge. In many patients, response to ADCs is followed by progression under treatment. To study this, we are actively generating PDX models from patients who initially responded to ADC therapy and later relapsed. In the AACR study, one such PDX was derived from a patient who had responded to mirvetuximab soravtansine before progressing. When re-challenged in vivo, this model showed resistance to treatment, suggesting that clinically acquired resistance mechanisms can be preserved in PDXs. Models like this provide powerful tools for studying resistance biology and evaluating nextgeneration therapies designed to overcome it. Why PDXs matter for ADC development Taken together, these findings show how PDX models can support ADC development beyond basic efficacy screening. When combined with molecular and bioinformatics analyses, they can help: Improve patient stratification beyond single biomarkers Reveal biological drivers of heterogeneous response Enable investigation of intrinsic and acquired resistance Support the development of next-generation ADCs and combination approaches As ADC pipelines continue to expand, integrating clinically representative PDX models early in development can improve translation and, ultimately, patient outcomes. Want to explore the full data? This blog highlights key insights, but the full dataset includes detailed efficacy results, pathway analyses, and figures that provide additional depth. Download our AACR 2026 poster to explore the complete results and analyses.
