Trends in Oncology
Understanding Sensitivity and Resistance to Pan-RAS(ON) Inhibition Across KRAS-Mutant Tumors
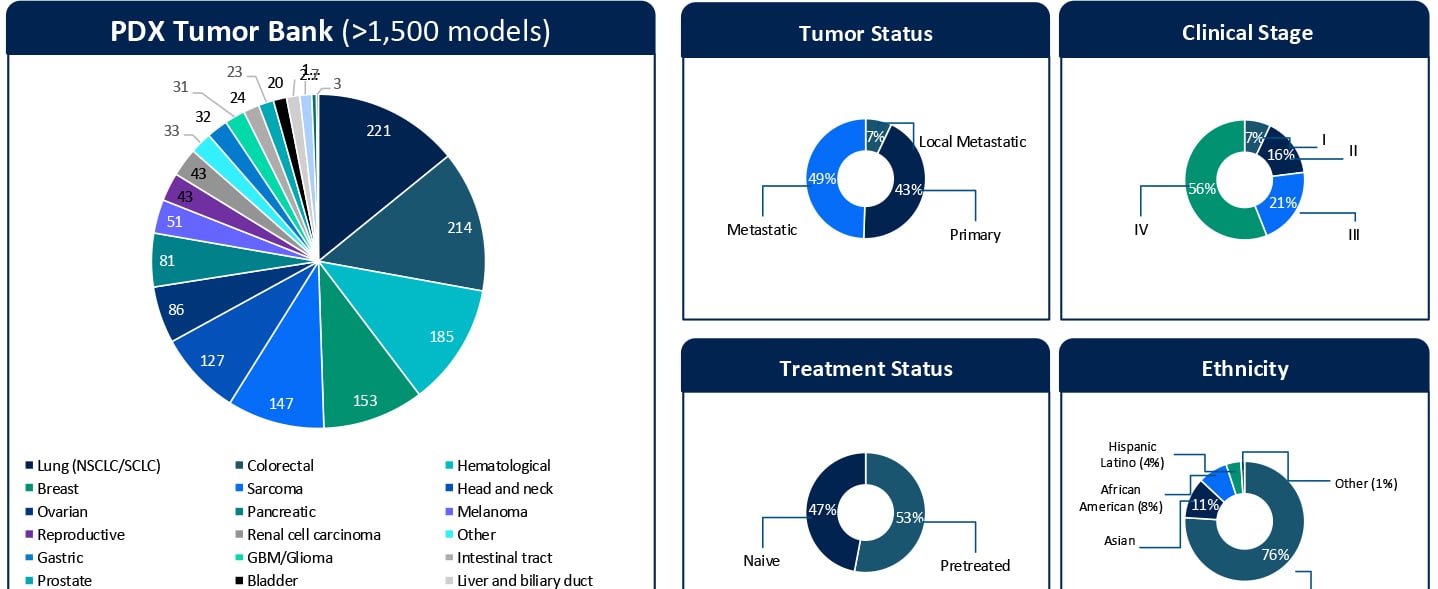
Understanding Sensitivity and Resistance to Pan-RAS(ON) Inhibition Across KRAS-Mutant Tumors KRAStargeted therapies are entering a new phase of clinical relevance. Daraxonrasib, a next-generation pan-RAS(ON) inhibitor designed to target a broad spectrum of KRAS G12X mutations, has shown impressive clinical activity. In a recent Phase III RASolute 302 trial for second-line metastatic pancreatic ductal adenocarcinoma (PDAC), daraxonrasib (RMC-6236) nearly doubled the median overall survival for patients to approximately 13.2 months, compared with 6.7 months for standard chemotherapy.1 These results highlight the rapid progress in this space. At the same time, clinical development is expanding KRAS-targeted therapies into earlier lines of therapy. Daraxonrasib is currently being evaluated in first-line settings, both as monotherapy and in combination approaches. While both clinical and preclinical data demonstrate activity across multiple tumor types, variability in response and the emergence of resistance remain key challenges that limit durable benefit. Additionally, the molecular determinants that define sensitivity, primary resistance, and acquired resistance remain incompletely understood. Addressing these gaps in our understanding of the molecular drivers of sensitivity and resistance is critical not only for patient selection but also for informing rational combination and sequencing strategies in an evolving treatment landscape. A Translational Platform to Study KRAS Biology in Context To better understand these response patterns, this study leveraged Champions Oncology’s low-passage, clinically annotated Patient-Derived Xenograft (PDX) Tumor Bank. With more than 1,500 models and extensive molecular and treatment history annotation, this platform captures both tumor heterogeneity and clinically relevant treatment context. Importantly, approximately half of the models represent pretreated disease, reflecting real-world patient populations. The translational relevance of this platform is supported by prior clinical correlation analyses performed using Champions’ low-passage PDX models. In a study by Izumchenko et al., treatment responses observed in these models were directly compared with clinical outcomes in matched patients across 92 individuals and 129 therapies.2 Strong concordance between the PDX and patient response was observed, with a positive predictive value of approximately 85% and a negative predictive value of approximately 91%. These findings establish Champions’ low-passage PDX models both as a tool for biological exploration and a clinically validated system for informing translational and drug development decisions. In this study, more than 50 KRAS-mutant PDX models were screened across non-small cell lung cancer, colorectal cancer, and pancreatic cancer (PDAC). The cohort included diverse KRAS alleles, co-occurring genomic alterations, and varying treatment histories, which allowed treatment response to be evaluated within a realistic molecular and clinical framework. Heterogeneous Responses Reflect Tumor-Specific Context Daraxonrasib treatment resulted in a broad spectrum of responses, ranging from strong disease regressions to progressive disease. Response variability was not solely explained by KRAS mutation status alone but was influenced by the broader genomic context of each tumor. For example, alterations such as MYC amplification were observed in both responding and non-responding models, suggesting that primary resistance is context-dependent and likely driven by multiple interacting genomic factors, rather than single genomic features. These findings highlight the importance of distinguishing between intrinsic resistance mechanisms and those that emerge under therapeutic pressure. Probing Primary Resistance in KRAS G12D Pancreatic Models Given the clinical relevance of KRAS G12D in pancreatic cancer, a focused analysis was performed in this subset. Differential gene expression analysis comparing responders and non-responders identified candidate markers associated with response, including MSLN and CYP24A1. While these findings are exploratory and based on a limited sample set, they provide an initial framework for stratifying tumors and generating hypotheses around mechanisms of intrinsic resistance. Ongoing work is expanding these analyses through deeper multi-omic integration, including whole exome sequencing (WES), RNA sequencing, proteomics, phosphoproteomics, and surface proteomics. These datasets will be interpreted in the context of Champions’ clinically annotated models, which include detailed treatment histories, to better understand the biological drivers of response and resistance and to support more robust biomarker development. Modeling Acquired Resistance Under Continuous Drug Pressure Selected PDX models were treated continuously beyond their initial response to investigate how resistance evolves over time. A particularly illustrative example is model CTG-2473, a KRAS G12D pancreatic cancer model derived from a tumor collected after first-line gemcitabine treatment. This model reflects a clinically relevant treatment sequence where patients, following standard chemotherapy, would be transitioned to KRAS-targeted therapies such as daraxonrasib. Therefore, treatment of CTG-2473 with daraxonrasib can model response and resistance in a post-chemotherapy setting that mirrors emerging clinical practice. Under continuous treatment pressure, this model demonstrated initial tumor regression followed by relapse, which is consistent with the development of acquired resistance observed in patients. This creates a powerful framework for translational studies. Experiments can be designed in the parental setting to evaluate therapeutic switching upon relapseto assess next-line agents or rational combination strategies. In parallel, Champions Oncology is actively developing a fully resistant subline through serial passaging under sustained drug pressure. This will enable deeper molecular characterization of acquired resistance mechanisms and provide a robust system for testing strategies for overcoming resistance. Together, these models establish a framework not only to observe resistance but also to actively model clinically relevant treatment sequencing scenarios. Why Clinically Annotated PDX Models Matter A defining strength of this work is the integration of molecular profiling, treatment response, and detailed clinical annotation within a single experimental system. Unlike conventional preclinical models, Champions’ PDX platform enables analysis of tumor behavior in the context of prior therapies, disease progression, and patient-specific treatment history. This is particularly important for targeted therapies such as KRAS inhibitors, where response and resistance are shaped by both molecular context and prior treatment. When combined with multi-omic datasets, including WES, RNA sequencing, proteomics, phosphoproteomics, and surface proteomics, these clinically annotated models provide a systems-level understanding of tumor biology. This supports the identification of candidate biomarkers and the development of mechanistically informed combination and sequencing strategies. Importantly, this integrated approach allows researchers to move beyond static response measurements tto capture how tumors dynamically adapt under therapeutic pressure and how these adaptations can be therapeutically targeted. From Mechanism to Strategy Taken together, these data demonstrate that response to pan-RAS(ON) inhibition is shaped by tumor-specific molecular context and dynamic adaptation under treatment. Understanding both primary and acquired resistance will be essential for translating KRAS-targeted therapies into durable clinical benefit. The full dataset, including detailed response profiles, genomic annotations, and resistance modeling, is presented in the accompanying poster. Download the Full Poster To explore the complete results, figures, and molecular analyses, including responder and non-responder comparisons and acquired resistance models, download the full poster here. References Daraxonrasib Demonstrates Unprecedented Overall Survival Benefit in Pivotal Phase 3 RASolute 302 Clinical Trial in Patients with Metastatic Pancreatic Cancer | Revolution Medicines [Internet]. Revolution Medicines. 2026. Available from: https://ir.revmed.com/news-releases/news-release-details/daraxonrasib-demonstrates-unprecedented-overall-survival-benefit Izumchenko E, Paz K, Ciznadija D, Sloma I, Katz A, Vasquez-Dunddel D, et al. Patient-derived xenografts effectively capture responses to oncology therapy in a heterogeneous cohort of patients with solid tumors. Annals of Oncology. 2017 Oct;28(10):2595–605.
